
STORY HIGHLIGHTS
- Researchers used stem cells to make organoids
- These organoids can be used to explore brain development
- Parts in organoids resemble brain regions in a 9-week-old human embryonic brain
- Embryonic and induced pluripotent stem cells yielded the same results
(CNN) — We’ve seen beating heart tissue, windpipes and bladders all grown from stem cells. Now researchers have taken another important step forward by growing mini brains from these programmable cells.
They’re not actually functioning brains — in the same way that a car with the engine on its roof or wheels on its hood isn’t a drivable vehicle — but the parts are there, and that’s an important scientific advancement, according to Juergen Knoblich, senior author of a new study on using stem cells to grow brain tissue.
Scientists have created what they are calling “cerebral organoids” using stem cells. These pea-sized structures are made of human brain tissue, and they can help researchers explore important questions about brain development and disorders that occur during these first stages of life.
The organoids, as described in the journal Nature, have components resembling those of a brain of a 9- or 10-week-old embryo, said lead study author Madeline Lancaster, a researcher at the Institute of Molecular Biotechnology at the Austrian Academy of Science in Vienna, at a press briefing Tuesday.
She and colleagues have created hundreds of these organoids.
At this early stage of human development, several key regions of the brain are already distinctive features, including the dorsal cortex, the ventral forebrain, the choroid plexus — which generates cerebrospinal fluid — and regions that resemble the midbrain and hindbrain. Lancaster and colleagues say they’ve identified some of those same regions in these new mini brains.
However, these regions did not naturally fall into place in the stem cell models the same way they would have in a normal brain.
“These different regions are not organized in the same kind of fashion that you would see in the developing embryo,” Lancaster said.
The organoids also lack certain features that human embryonic brains at 9 weeks do have: most importantly, the cerebellum, which is involved in motor movement. Also, the hippocampus, a seahorse-shaped structure crucial for memory, was rarely detected in these brain-like structures.
Researchers used human embryonic stem cells and induced pluripotent stem cells (IPS cells) for this research. Both embryonic stem cells and IPS cells have the ability to turn into any part of the body. But embryonic stem cells are very controversial because in the process of retrieving them for research, the 4- or 5-day-old embryo they are taken from is destroyed. IPS cells don’t come with the same controversy because scientists take a cell — typically a skin cell — then coax it using a chemical bath to revert to a state that resembles a developing embryo.
There did not appear to be an obvious difference between organoids derived from embryonic stem cells and those produced from IPS cells, said Knoblich, also of the Austrian Academy of Science.
Study authors found variability in the organoids they generated; occasionally some of the brain regions they were studying failed to appear.
Lancaster attempted to direct the development of these regions in some of the organoids by applying growth factors, substances that promote the proliferation of cells. Surprisingly, when she tried to grow the mini brains with more dorsal cortex tissue, the resulting structures had less of this tissue than the organoids that had developed on their own.
“We actually think that the cross-talk between these different regions — the communication between these different brain regions within the organoids — is really important for each individual region’s development,” she said.
The researchers used this model to look at a neurodevelopmental disease called microcephaly, a disorder in which the size of the brain is reduced. The brain region they were most interested in exploring, the dorsal cortex, is the region most highly impacted by this disease.
Researchers grew some of these organoids using cells from a patient who had a genetic form of microcephaly, and compared them with the mini brains derived from healthy participants’ cells.
In the organoids made from the microcephaly patient’s cells, it appeared that more stem cells had been turned into neurons — a process called differentiation — than in the mini brains derived from healthy patients’ cells. This suggests that in people with this condition, neurons prematurely differentiate, which could be the mechanism behind this form of the disease, said Oliver Brustle at the Life & Brain Centre at University of Bonn, in an accompanying article in Nature.
This research builds on other studies that have attempted to model brain tissue from stem cells. A 2008 study showed that mouse embryonic stem cells could be coaxed into producing “waves” of neurons. A different research group showed in 2012 that primitive eye structures and stratified retinas could form from embryonic stem cells taken from both mice and humans. Study authors said they have no intention of growing a full-sized human brain.
“It is very clear that our system is not optimized for generating an entire brain, and that is also in no way our goal,” Knoblich said.
As for growing a brain structure from stem cells that’s capable of conscious thought, Knoblich said this would likely not be possible, or desirable.
Although the organoids are an important step forward, the researchers are nowhere near being able to model circuits found in the functional central nervous system. Moreover, Knoblich said, sensory input is required for such functional circuits to form. A classical experiment showed that the optic cortex will not organize properly if it does not have input from an eye, he said.
Knoblich is also pessimistic about the idea of growing brain structures from stem cells with the intention of replacing faulty ones in human patients. The brain is so complex, and its regions so intimately integrated, that it would be difficult to repair any specific part through substitution.
A more promising possibility, he said, would be to put the stem cells directly into the patient and let them organize themselves. But the future of this line of research is still unknown.
Brustle, who was not involved in this research, called the study “remarkable” and noted that it “clearly puts neural aggregation cultures on the map of research tools for both developmental biology and biomedicine.”
That’s a lot from a little tissue.
Follow Elizabeth Landau on Twitter and Google+ .
Jump to original:
Scientists grow brains from cells
The post Scientists grow brains from cells appeared first on Arne Ruhnau News.
via Arne Ruhnau News http://arneruhnau.com/scientists-grow-brains-from-cells/
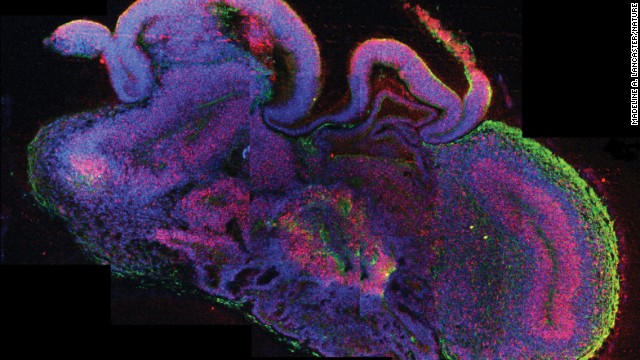 Scientists have created what they are calling “cerebral organoids” using stem cells. These pea-sized structures are made of human brain tissue, and they can help researchers explore important questions about brain development and disorders that occur during these first stages of life. Click through to learn more about stem cell research.
Scientists have created what they are calling “cerebral organoids” using stem cells. These pea-sized structures are made of human brain tissue, and they can help researchers explore important questions about brain development and disorders that occur during these first stages of life. Click through to learn more about stem cell research.  Stem cells have the potential to become many different kinds of cells, and can renew themselves through cell division. Scientists view stem cells as a possible gateway to curing many medical conditions, from Parkinson’s disease to diabetes. Stem cells are viewed on computer here at UConn Health Center in 2010.
Stem cells have the potential to become many different kinds of cells, and can renew themselves through cell division. Scientists view stem cells as a possible gateway to curing many medical conditions, from Parkinson’s disease to diabetes. Stem cells are viewed on computer here at UConn Health Center in 2010.  A closeup of a microscope slide taken in 2000 at the Reproductive Genetics Institute’s Chicago laboratory shows transplanted stem cells taken from the umbilical cord blood of a baby named Adam Nash. Adam’s sister Molly has a genetic disease called Franconi Anemia. Their parents wanted to have a child who could be a stem cell donor for Molly. Using in vitro fertilization, doctors created embryos and then tested them for the genetic disease. They chose one that did not have the disorder, which grew into baby Adam. Molly received a stem cell transplant from stem cells from Adam’s umbilical cord. Both children are alive today.
A closeup of a microscope slide taken in 2000 at the Reproductive Genetics Institute’s Chicago laboratory shows transplanted stem cells taken from the umbilical cord blood of a baby named Adam Nash. Adam’s sister Molly has a genetic disease called Franconi Anemia. Their parents wanted to have a child who could be a stem cell donor for Molly. Using in vitro fertilization, doctors created embryos and then tested them for the genetic disease. They chose one that did not have the disorder, which grew into baby Adam. Molly received a stem cell transplant from stem cells from Adam’s umbilical cord. Both children are alive today.  In 1998, then-President Bill Clinton requested a National Bioethics Advisory Commission to study the question of stem cell research.
In 1998, then-President Bill Clinton requested a National Bioethics Advisory Commission to study the question of stem cell research.  In 2000, The National Institutes of Health issued guidelines for the use of embryonic stem cells in research, specifying that scientists receiving federal funds could use only extra embryos that would otherwise be discarded. President Clinton approved federal funding for stem cell research but Congress did not fund it. Above, a Cell Expansion System which is used to grow cells is seen during the 2010 World Stem Cell Summit in Detroit.
In 2000, The National Institutes of Health issued guidelines for the use of embryonic stem cells in research, specifying that scientists receiving federal funds could use only extra embryos that would otherwise be discarded. President Clinton approved federal funding for stem cell research but Congress did not fund it. Above, a Cell Expansion System which is used to grow cells is seen during the 2010 World Stem Cell Summit in Detroit.  In August 2001, then-President George W. Bush announced he would allow federal funding for about 60 existing stem cell lines created before this date. Above, a human stem cell colony, which is no more than 1mm wide and comprises thousands of individual stem cells, grows on mouse embryonic fibroblast in a research laboratory in September 2001.
In August 2001, then-President George W. Bush announced he would allow federal funding for about 60 existing stem cell lines created before this date. Above, a human stem cell colony, which is no more than 1mm wide and comprises thousands of individual stem cells, grows on mouse embryonic fibroblast in a research laboratory in September 2001. 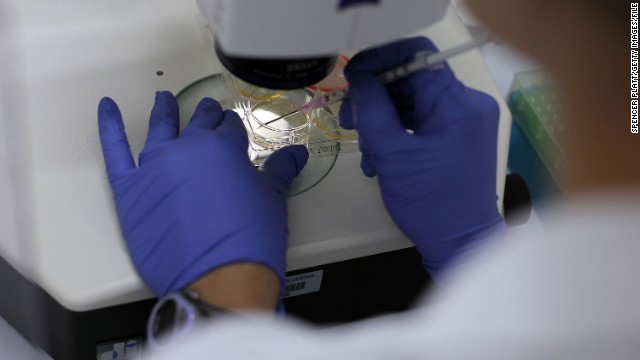 In 2005, Connecticut and Illinois designated state funds to support stem cell research in their states. Above, a woman works on stem cells at the University of Connecticut’s Stem Cell Institute at the UConn Health Center in August 2010 in Farmington, Connecticut.
In 2005, Connecticut and Illinois designated state funds to support stem cell research in their states. Above, a woman works on stem cells at the University of Connecticut’s Stem Cell Institute at the UConn Health Center in August 2010 in Farmington, Connecticut.  In March 2009, President Barack Obama signed an executive order that removed restrictions on embryonic stem cell research. His action overturned an order approved by President George W. Bush in August 2001 that barred the National Institutes of Health from funding research on embryonic stem cells beyond using 60 cell lines that existed at that time. Above, Obama signs the order.
In March 2009, President Barack Obama signed an executive order that removed restrictions on embryonic stem cell research. His action overturned an order approved by President George W. Bush in August 2001 that barred the National Institutes of Health from funding research on embryonic stem cells beyond using 60 cell lines that existed at that time. Above, Obama signs the order.  In November 2010, William Caldwell, CEO of Advanced Cell Technology, told CNN that the FDA had granted approval for his company to start a clinical trial using cells grown from human embryonic stem cells. The treatment would be for an inherited degenerative eye disease. Above, dozens of packages containing frozen embryonic stem cells remain in liquid nitrogen in a laboratory, at the University of Sao Paulo’s human genome research center, in Sao Paulo, Brazil, in March 2008.
In November 2010, William Caldwell, CEO of Advanced Cell Technology, told CNN that the FDA had granted approval for his company to start a clinical trial using cells grown from human embryonic stem cells. The treatment would be for an inherited degenerative eye disease. Above, dozens of packages containing frozen embryonic stem cells remain in liquid nitrogen in a laboratory, at the University of Sao Paulo’s human genome research center, in Sao Paulo, Brazil, in March 2008.  In May 2011, stem cell therapy in sports medicine was spotlighted after New York Yankee pitcher Bartolo Colon was revealed to have had fat and bone marrow stem cells injected into his injured elbow and shoulder while in the Dominican Republic. Above, Colon pitches against the Boston Red Sox on in May 2011.
In May 2011, stem cell therapy in sports medicine was spotlighted after New York Yankee pitcher Bartolo Colon was revealed to have had fat and bone marrow stem cells injected into his injured elbow and shoulder while in the Dominican Republic. Above, Colon pitches against the Boston Red Sox on in May 2011.  In February 2012, early research published by scientists at Cedars-Sinai Medical Center and Johns Hopkins University showed that a patient’s own stem cells can be used to regenerate heart tissue and help undo damage caused by a heart attack. It is the first instance of therapeutic regeneration. Above, fluid is removed from the knee of a patient to collect adult stem cells by at a clinic in Broomfield, Colorado.
In February 2012, early research published by scientists at Cedars-Sinai Medical Center and Johns Hopkins University showed that a patient’s own stem cells can be used to regenerate heart tissue and help undo damage caused by a heart attack. It is the first instance of therapeutic regeneration. Above, fluid is removed from the knee of a patient to collect adult stem cells by at a clinic in Broomfield, Colorado.  In October 2012, Sir John Gurdon and Shinya Yamanaka were awarded the Nobel Prize for Physiology or Medicine for discovering how to make pluripotent stem cells. They both showed that cells could be reprogrammed after they had already specialized. This changed scientists’ understanding of how cells and organisms develop. Above, Sir Gurdon speaks at a press conference after being awarded the Prize.
In October 2012, Sir John Gurdon and Shinya Yamanaka were awarded the Nobel Prize for Physiology or Medicine for discovering how to make pluripotent stem cells. They both showed that cells could be reprogrammed after they had already specialized. This changed scientists’ understanding of how cells and organisms develop. Above, Sir Gurdon speaks at a press conference after being awarded the Prize. 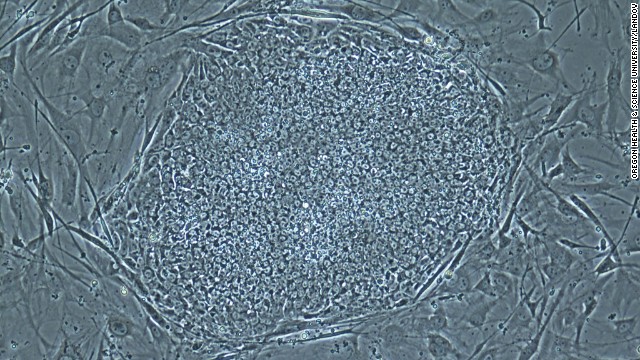 On May 16, 2013, scientists announced that they had, for the first time, produced embryos using skin cells, and then used the embryos to make stem cell lines. This technique resembles what was used in cloning Dolly the sheep, but the earlier technique could not have led to a fully-cloned human baby. Above, a photo provided by the Oregon Health & Science University shows a stem cell colony produced from human skin cells.
On May 16, 2013, scientists announced that they had, for the first time, produced embryos using skin cells, and then used the embryos to make stem cell lines. This technique resembles what was used in cloning Dolly the sheep, but the earlier technique could not have led to a fully-cloned human baby. Above, a photo provided by the Oregon Health & Science University shows a stem cell colony produced from human skin cells.  On Tuesday, August 5, the world’s first stem cell burger was cooked and eaten in London. The brainchild of Maastricht University’s Mark Post, the burger was made of 20,000 small strands of meat grown from a cow’s muscle cells and took three months to create and cost $330,000 to develop.
On Tuesday, August 5, the world’s first stem cell burger was cooked and eaten in London. The brainchild of Maastricht University’s Mark Post, the burger was made of 20,000 small strands of meat grown from a cow’s muscle cells and took three months to create and cost $330,000 to develop. 









No comments:
Post a Comment